
Combining high-resolution structural views with plaque compositional analysis to deliver unprecedented treatment insights.
The Makoto® Intravascular Imaging System is an FDA-cleared imaging system indicated for the detection of lipid core plaque (LCP) and the identification of plaque and patients at increased risk of MACE. The system automatically quantifies the total lipid core in regions of interest and calculates a plaque’s Lipid Core Burden Index (LCBI). Multiple studies have confirmed that the LCBI number can be used to evaluate patient and plaque risk of MACE, gauge the effectiveness of current therapies, and adjust future treatment plans.
The Makoto® Intravascular Imaging System brings you valuable insights that no other system can.
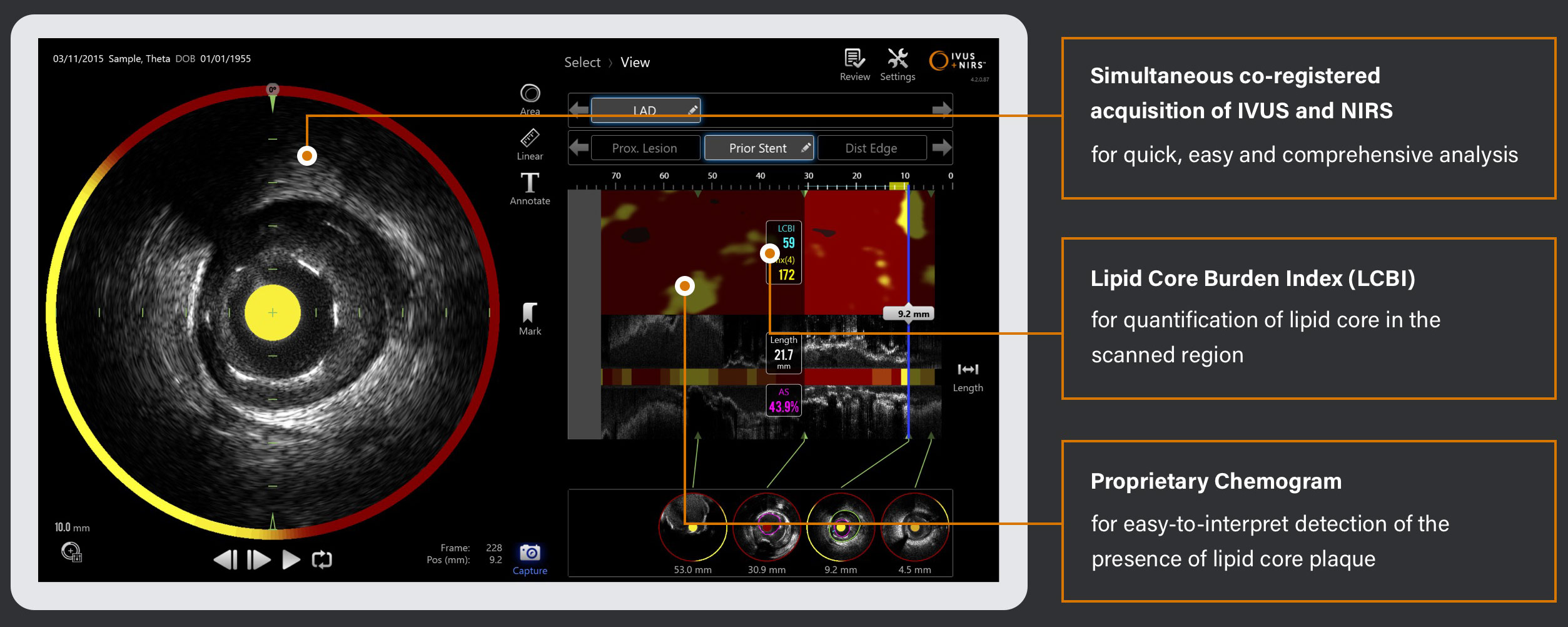
Multiple studies have demonstrated that assessment of the coronary artery with the Makoto® IVUS+NIRS Intravascular Imaging System is unmatched in its ability to identify the risk of MACE. The results of the landmark LP study provided clinical evidence that intravascular NIRS imaging can accurately identify both vulnerable patients and vulnerable plaques that are at significantly higher risk for subsequent NC-MACE during a 24-month period.
Assessment of Coronary Near Infrared Spectroscopy Imaging to Detect Vulnerable Patients and Vulnerable Plaques
Assessment of Coronary Near Infrared Spectroscopy Imaging to Detect Vulnerable Patients and Vulnerable Plaques
US Customers: Click here to proceed to Nipro.com
References
© 2026 Infraredx™, Inc. All Rights Reserved Powered by Bloom Creative